
Robotic surgery for urinary fistulae in females: a narrative review
Introduction
Fistulae are abnormal communications between two epithelial surfaces (1). They are debilitating conditions that result in social, psychological, and physical morbidity and decreased quality of life (2-4).
About 2–3 million women worldwide have an untreated fistula. Approximately 95% of fistulae cases occur in third-world countries as a result of obstructed labor (5). In the developed world, non-obstetric causes such as surgery, infections, congenital anomalies, malignancy, and radiotherapy are more common (2,5-9).
Fistulae were initially described by Rufus of Ephesus in 200 A.D. Management has evolved to include more minimally invasive approaches (10).
With the outcome of minimally invasive approaches, the morbidity of surgical procedures decreased with reduced hospital stay, shorter recovery time, lower complication rates, pain medication requirements, estimated blood loss, and better cosmesis. Furthermore, the use of intravenous indocyanine green allows the visualization of tissue perfusion while operating. It is particularly helpful in identifying the ureter and delineating the ureteral stricture margins in cases where ureteral reconstruction is needed (11-13).
The development of robotic surgical systems was created to overcome the limitations of laparoscopy. Robotic surgery eliminates the fulcrum effect, which disrupts the natural hand-eye coordination, provides three-dimensional (3D) visualization with a wider range of arm movement and a more precise dissection, with a shorter surgical learning curve and easier intracorporeal suturing while providing an ergonomic position for the surgeon. Conversely, robotic surgery represents higher costs due to additional surgical training, expensive maintenance equipment, and increased operating room setup time (14-16).
The main objective of this narrative review was to discuss relevant aspects of the vesicovaginal fistula (VVF), vesicouterine fistula (VUF), and ureterovaginal fistula (UVF) and discuss their robotic repair. We present this article in accordance with the Narrative Review reporting checklist (available at https://gpm.amegroups.com/article/view/10.21037/gpm-24-13/rc).
Methods
A narrative review was conducted utilizing a comprehensive search to identify studies discussing the robotic management of female urinary fistulae. Publications were selected based on their relevance to the chosen content. Data from all types of studies were included without restrictions on article type. Only articles in English and Spanish were considered. Searches were performed on 12/12/2020, 04/25/2023, and 09/10/2023 in the electronic database PubMed (1978 to 2023), using free text terms and keywords (see Table 1). After removing duplicate studies, the review included seventeen case reviews, eighteen case series, thirty-one narrative reviews, one retrospective cohort study, one randomized controlled trial, and four systematic reviews. Additionally, secondary searches were conducted based on papers found in reference lists to supplement the primary literature search and support the description of basic concepts. The themes identified from the literature review were categorized into subheadings for this review.
Table 1
| Item | Specifications |
|---|---|
| Search dates | 12/12/2020, 04/25/2023, 09/10/2023 |
| Database searched | PubMed |
| Search keywords | “Female genitourinary fistula”, “vesicovaginal fistula”, “vesicouterine fistula”, “ureterovaginal fistula”, “robotic management urinary fistula” “robotic fistula management”, “ICG robotic surgery”, “ICG fistula” |
| Timeframe | 1978–2023 |
| Inclusion criteria | All type of studies written in English or Spanish were included |
| Selection process | Relevant papers were identified by the first author based on the relevance of their content, which guided secondary searches and the inclusion of additional citations from reference lists |
ICG, indocyanine green.
Diagnosis
The diagnosis of urinary fistulae should include a detailed history with physical examination, cystoscopy, or vaginoscopy, and imaging with either magnetic resonance imaging (MRI) or computerized tomography (CT) is recommended to provide detailed anatomic information for surgical planning (7,17-19).
Beginning with the history and physical examination, the manifested signs and symptoms of urinary tract fistulae will vary depending on the size and anatomical location of the lesions and the organs involved. Timing will differ depending on the cause (17,20). Post-operative fistulae present as early complications approximately ten days after the surgery. These may occur when the bladder, ureters, or urethra has unrecognized damage that causes necrosis and, eventually, the formation of a fistula. In contrast, radiation-induced fistulae frequently occur years after treatment. This is due to progressive endarteritis, hypoxia, fibrosis, and necrosis (6,8,21).
VVFs are characterized by continuous urinary leakage from the vagina, which can vary in severity depending on the size of the lesion. This leakage can range from barely noticeable to causing significant distress to the patient (6,22). In contrast, UVFs result in constant urinary leakage and periodic filling cycles of the bladder due to the unaffected contralateral ureter (7). The diagnosis of VVF can be established in a clinical setting using the “Tampon test”. This test involves filling the bladder with a diluted solution of methylene blue, inserting a tampon into the vagina, and instructing the patient to ambulate. Additionally, the dual-dye tampon test may be employed when there is suspicion of a concurrent UVF. This test involves the administration of Pyridium orally two hours before conducting the tampon test. The presence of both orange and blue dye on the tampon confirms the presence of a UVF alongside the vesicovaginal fistula. Care should be taken if the patient has another cause of incontinence, such as stress incontinence, as the placement of the tampon is crucial for accurately performing the test (23).
Assessing the size, location, and surrounding tissue availability is important in directing management decisions. Identifying pertinent comorbidities, presence of concomitant fistulae, strictures, and sphincter incompetence should be accomplished through thorough history-taking, physical examination, and, if warranted, imaging studies. A comprehensive evaluation, including bimanual and speculum examinations, can provide necessary insights for management planning (7,17,20).
Cystoscopy and vaginoscopy are performed to evaluate the location, complexity, and tissue quality surrounding the fistulae. When clinical suspicion of a malignant origin arises, an endoscopic biopsy of the fistula tract becomes imperative. Early determination of the presence of urologic, gynecologic, colorectal, or other will influence the approach to fistulae management.
Imaging can be a helpful adjunct for both diagnosis and surgical planning. When uncertain, techniques such as voiding cystourethrograms or retrograde urethrograms can confirm the diagnosis and offer detailed insights into the anatomical location and size of the fistula. In general, a retrograde pyelogram is used intraoperatively to determine the location of the fistulae when attempting ureteral stent placement for conservative treatment. Additionally, upper tract evaluation, primarily conducted through computerized tomography urography (CTU), is recommended in cases of postsurgical VVF due to a 12% incidence of ureteral injury or UVF. This is particularly relevant when the uterus is present or when the tampon test results are indeterminate. CTU can achieve a sensitivity of up to 70% and a specificity of up to 100% (7,20,23-26).
Treatment overview
Treatment of genitourinary fistulae depends on the size, location and underlying cause. A small subset can resolve with conservative management (urinary drainage), but more commonly, fistulae are treated surgically (21). This narrative review will focus on the robotic management of female genitourinary fistulae.
Principles of surgical management for genitourinary fistulae
Patients may require surgery for larger, more complicated fistulae or if they have failed conservative management. The optimal timing for surgery varies depending on several factors (7). It is widely recognized that the highest likelihood of successful fistulae closure occurs during the initial surgical attempt, with each subsequent attempt carrying an increased risk of failure. However, no consensus exists regarding the optimal interval between onset symptoms and fistulae repair (4).
In cases of iatrogenic fistulae detected promptly after surgery, immediate repair is hypothesized to be advantageous, as delaying intervention may lead to loss of tissue planes and fibrosis. Conversely, in instances of delayed recognition of genitourinary fistulae or those arising secondary to radiotherapy, it is theorized that waiting approximately three months allows for the absorption of granulation tissue and edema before surgical intervention (4,6).
There are several general surgical principles essential in ensuring successful surgical repair. These include good visualization, adequate exposure, careful dissection, healthy margins, and watertight closure with suture lines that do not overlap (4,7,27-29).
When closure is accomplished using healthy, well-vascularized tissue, success rates tend to be higher. However, there are instances where tissue mobilization for staggering suture lines is not feasible, and there is a lack of healthy intervening local tissue for closure, increasing the risk of breakdown and fistula recurrence. In such scenarios, the utilization of an interposition tissue flap is recommended to augment local metabolic and immune functions and, at the same time, act as a barrier to the suture lines. There are a wide variety of tissue types that can be used, such as omentum, peritoneum, muscle, and vaginal wall (7).
The management of specific types of genitourinary fistulae is detailed below.
VVFs
A VVF is an abnormal communication between the bladder and the vagina. VVFs are the most commonly acquired urinary tract fistulae, with an incidence between 0.3% and 2% of women worldwide. In developed countries, gynecologic surgeries, such as hysterectomies, are the most common cause, accounting for about 75% of the VVFs cases (10,17,22,26,30-36).
The management of VVFs depends on several factors, including the location, the quality of the tissue, the size of the vaginal space, the number of previous surgical attempts, and the surgeon’s expertise. Abdominal surgery is typically preferred for fistulae larger than 3 cm, cases with a narrow vaginal space, fistulae resulting from radiation therapy, supratrigonal fistulae, or those located near the ureters (22,30,31). The success rate for transabdominal surgical correction ranges from 75% to 97%. In contrast, the transvaginal approach is generally suitable for small, non-recurrent, non-radiated, and lower-location fistulae, with success rates between 80% and 90% (29,30,37,38).
The robotic-assisted abdominal repair offers two approaches with different benefits and risks: transperitoneal transvaginal and transvesical. The transperitoneal transvaginal approach minimizes bladder incisions and reduces the recurrence rate and irritative voiding symptoms with success rates of 98–100% (29,30) (Figure 1). The transvesical approach allows direct visualization of ureteral orifices and wide dissection of the fistulous tract but can be associated with decreased bladder capacity, recurrent urinary tract infections, and detrusor dysfunction secondary to the cystostomy. The transvesical approach has success rates between 80–100% (4,39) (Figures 2-4). Given that no randomized control trials to date compare both techniques, the best technique is yet to be determined, and the most critical factor is still surgeons’ preference (22,29).


The robotic-assisted transperitoneal transvaginal should begin with a cystoscopy to confirm the fistula orifice and placement of stents through the fistula tract for better identification (Figure 3). The use of a vaginal occluder is recommended to keep pneumoperitoneum (30,39).
Laparoscopic entry can be achieved at the umbilicus according to surgeon preference. An 8-mm port is then inserted with a 30° lens, recommended for better visualization at the colpotomy site, followed by two 8-mm ports parallel to the right and left pararectal lines, and a fourth 8-mm port is placed on the right side iliac crest, above the iliac crest, but below ports 2 and 3. Lastly, the air seal port is located on the left upper quadrant (Figure 5). After all port placement, the robot is docked (30,40).
In the presence of adhesions, surgery should begin with adhesiolysis. To ensure successful surgical repair, an omental flap can be created and positioned to test the future placement site between the vagina and bladder, as shown in Figure 6. The vagina is identified and incised, guided by the stent previously placed at the fistula tract. The fistula tract is excised, ensuring only healthy, viable tissue remains for repair (Figures 2-4). The bladder and vagina are dissected, and the vaginal opening is repaired, avoiding overlap between the suture lines, using running stitches horizontally with absorbable braided suture (40-43).
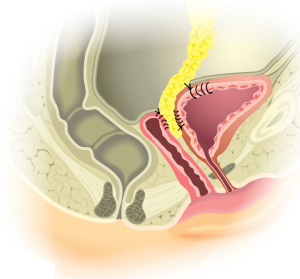
Cystorraphy is approximated in two layers following a vertical direction with braided absorbable sutures. The closure is then tested with methylene blue (44). The selected flap is then interposed and anchored between the bladder and the vagina (Figure 6). If omentum is unavailable due to prior surgery, a peritoneal flap can be used as an alternative (7,30,34,39,45).
Finally, a cystoscopy is performed to identify the ureteral orifices.
The transvesical approach adapts the O’Connor procedure initially described by O’Connor as an open technique (46-48). Like the transperitoneal approach, the first step is to identify the fistulae and ureters with a different color ureteral catheter and double-J stents (Figure 3). Port placement will be in a fifth-port transperitoneal configuration (Figure 5). The patient is then positioned in a steep Trendelenburg position. A longitudinal cystotomy is executed with monopolar robotic scissors positioned just above the fistulous tract (Figures 2-4) (41,49). Concurrently, a vaginal pack will be placed to maintain the pneumoperitoneum.
The cystotomy is advanced until the borders of the fistulous tract are excised, elucidating clearer communication between the two organs (Figures 3,4). The anatomical plane between the bladder and the vagina is dissected, followed by closure of the vagina using a transverse running locking fashion (21,34,41,49,50).
After the surgical repair is completed, the patient is repositioned horizontally. The previously harvested flap is placed between the suture lines (Figure 6).
The closure of the bladder is performed in a running fashion, initiating at the apex of the cystostomy. Subsequently, to ensure a watertight seal, 180 mL of dilute methylene blue is instilled, and the closure is inspected for any signs of leakage. In the event of leakage, an additional simple interrupted suture may be applied (10,21,34,50-52).
No matter the approach, the postoperative care will be standard. The ureteral catheters are later removed post-operatory day two or three, and an 18-Fr urethral catheter will stay in place until postoperative day 14th. Routinely, a cystogram will be performed 14 days after surgery, and if no residual fistula is observed, the catheter is removed (21,30,34).
VUFs
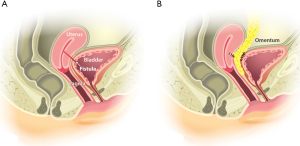
A VUF is an abnormal communication between the bladder and the uterus (39,53) (Figure 7A).
These are rare, comprising 2–9% of all urogenital fistulae. Most (83–88%) are associated with cesarean sections, although occurrences have been reported in women with congenital anomalies and postmenopausal women (53-58).
Risk factors for VUFs include inadequate dissection of the bladder from the lower uterine segment at the time of hysterectomy, severe labor dystocia, operative deliveries such as forceps or vacuum extraction, previous cesarean section, causing gradual tissue damage and scarring of the uterus and the base of the bladder, predisposing to fistula formation (53).
VUF presentation may vary, encompassing cyclic hematuria, amenorrhea, and infertility with or without urinary incontinence, known as Youssef syndrome (55,56,58).
The flow of urine or menstrual blood through the fistula is determined by the pressure difference between the uterine and bladder cavities. In most cases, intrauterine pressure exceeds bladder pressure, except during urination, which accounts for menouria and intermittent urinary incontinence observed in many patients with VUF (55).
Radiographic evaluation includes a cystogram and hysterogram to identify the fistula and location. A hysterogram is particularly useful in cases of menouria, as it highlights the fistula by showing contrast leaking (55).
Surgical treatment may differ depending on the patient’s future fertility desires. If there is completion of fertility, hysterectomy and closure of the bladder opening will be the best treatment option. On the other side, successful term pregnancies have been documented following uterine preservation in VUF repair cases (59). Vaginal repair is not commonly pursued due to limited accessibility to VUFs via this approach (55,56).
Our recommendation is to perform a transabdominal robotic repair. After initial cystoscopy with catheterization of both ureters with 5-Fr catheters and identification of the fistula with a different color catheter, placement of robotic ports, and docking. Dissection of the vesicouterine space is performed, followed by cystotomy at the bladder dome. The fistula tract is then excised, ensuring the mobilization of tissue layers to enable tension-free closure.
The uterus and bladder repair are performed with absorbable sutures in two layers in a running fashion. The use of interposition tissues at the repair site is recommended (Figure 7B). At the end of the repair, we recommend to backfill the bladder to confirm a watertight repair (54,60).
UVF
UVFs are abnormal communications between the ureter and the vagina (23,30,61) (Figure 8A). They are uncommon, with an estimated incidence of 0.16% occurring after ureteral injury during pelvic surgery (18,30). Furthermore, retained pessaries, stone fragments after shock wave lithotripsy, radiation, trauma, or infection represent other causes of UVF (14,46,49).

Delayed management of UVFs can lead to ureteral strictures, infections, and compromised renal function. If any secondary conditions are suspected, preoperative imaging should be obtained to aid in further evaluation and surgical planning (61-63).
Ureteral stenting should be attempted as first-line therapy. When it fails, there is a contraindication for its placement, or in the case of complex fistulae, procedures such as ureteroneocystostomy (Figure 8B), Boari flap, or psoas hitch can be performed using a minimally invasive approach (18,23,63,64). The repair can be performed depending on the characteristics of the UVF. In cases where periureteral inflammation is minimal and only a short segment of the ureter is implicated in the fistulae, a primary ureteral repair or a ureteroureterostomy may be considered using interrupted stitches of absorbable suture instead of ureteral reimplantation (65,66).
During a ureteroneocystostomy procedure, the ureter is identified and meticulously dissected down to the level of the fistula, transected and spatulated, ensuring preservation of the periureteral adventitia, and then sutured to the bladder with interrupted stitches of 5.0 monofilament (Figure 8B). To reinforce the ureteral attachment, the ureteral adventitia is often sutured to the bladder peritoneum (37).
In situations where there are ureteral defects or complete transection of the ureter, the ureteral repair should be deferred until the vaginal tract is repaired. A psoas hitch procedure is considered when a primary ureteroureterostomy or ureteroneocystostomy cannot be performed without tension (67,68). During the psoas hitch procedure, the ureter is transected proximal to the fistula. The bladder is then mobilized and secured to the psoas fascia using a nonabsorbable suture, with special attention to identifying the genitofemoral nerve. Cystotomy is performed at the bladder dome, and the ureter is spatulated and anastomosed in a tension-free repair. A stent-over-wire is inserted through the anastomosis and the procedure is completed after stent placement (65,66). Depending on the surgeon’s preference, a round Jackson Pratt drain can be placed in the pelvis for monitoring (66).
Post-operative management
When placed, the Jackson-Pratt drain is typically removed once the 24-hour output is less than 50 mL (22,39).
The Foley catheter is kept for a minimum of 14 days for postoperative management of VVF, VUF, and UVF. It can be maintained longer if the tissue quality is noted to be deficient while performing the surgery. Conversely, 7-day bladder catheterization can be considered, supported by evidence of no significant increased risk when removed earlier (22,39,69,70). A retrograde cystogram should confirm no contrast extravasation before catheter removal in vesicovaginal and vesicouterine cases (22,39).
If double-J stents were placed during the surgery, they might be left in place if the fistula or reconstruction was near the ureteral orifices (39). For UVF repairs, leaving stents in place for at least 4–6 weeks is recommended (38).
Pelvic rest is recommended for at least six weeks, avoiding vaginal intercourse, tampon usage, or douching for any fistulae repair (37,39).
Follow-up
Available literature reports a wide range of recommended follow-ups, from 1 to 12 months. It is assumed that the recurrence of a fistula is quick; in theory, a 3-month period is sufficient. However, ureteral strictures develop within 1 year following repair, with 0.3% to 3.4% after ureteroneocystostomy. Hence, the importance of assessing for stricture formation with CT urogram 3 months after stent removal up to 1 year (23,39,71). For the cases where the bladder is involved, a cystoscopy at 12 months can be performed to verify successful repair (39).
Associated issues
The use of anticholinergics is strongly recommended for the management of the irritative symptoms caused by prolonged urinary catheterization (37).
Between 10% and 55% of women who undergo successful fistulae closure experience stress incontinence afterward. Significant risk factors for post-fistulae closure incontinence include bladder, neck, or proximal urethral fistulae. Reported methods to address post-fistulae repair urethral incontinence encompass urethral plugs, urethropexy, urethral bulking, or sub urethral slings utilizing autologous fascia, synthetic mesh, or less commonly, the pubococcygeal muscle or biologic graft (72).
Prevention
Strategies to prevent fistulae formation should be related to address their etiology. In the United States, most fistulae arise during hysterectomy procedures following bladder or ureteral injury. Enhanced intraoperative recognition and prompt repair of genitourinary trauma can significantly reduce the incidence of fistulae formation. Improved detection is facilitated by the routine use of concurrent cystoscopy, which is increasingly becoming standard practice, especially following minimally invasive procedures (37).
Limitations
This review article’s limitations include possible gaps in literature search practices. Additionally, few studies report long-term outcomes for patients following genitourinary fistulae repair.
Conclusions
Fortunately, female urinary fistulae are uncommon, but when they do occur, they profoundly impact women’s quality of life. Surgical management is necessary in complicated cases and when conservative measures fail. When available, robotic surgery can aid in repairing the more difficult and complex issues, given its advantages of offering a minimally invasive approach with greater dexterity and a tridimensional view.
Acknowledgments
None.
Footnote
Reporting Checklist: The authors have completed the Narrative Review reporting checklist. Available at https://gpm.amegroups.com/article/view/10.21037/gpm-24-13/rc
Peer Review File: Available at https://gpm.amegroups.com/article/view/10.21037/gpm-24-13/prf
Funding: None.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://gpm.amegroups.com/article/view/10.21037/gpm-24-13/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Dellis AE, Delaportas D, Nastos K, et al. Female urogenital fistulas. Hellenic Urology 2017;29:19-23.
- D'Elia C, Curti P, Cerruto MA, et al. Large Urethro-Vesico-Vaginal Fistula due to a Vaginal Foreign Body in a 22-Year-Old Woman: Case Report and Literature Review. Urol Int 2015;95:120-4. [Crossref] [PubMed]
- Vaso M, Betschart C, Egger H, et al. Surgical technique of a recurrent post-radiation vesicovaginal fistula with a small intestine graft. Arch Gynecol Obstet 2015;292:485-8. [Crossref] [PubMed]
- Ramphal SR. Laparoscopic approach to vesicovaginal fistulae. Best Pract Res Clin Obstet Gynaecol 2019;54:49-60. [PubMed]
- Cromwell D, Hilton P. Retrospective cohort study on patterns of care and outcomes of surgical treatment for lower urinary-genital tract fistula among English National Health Service hospitals between 2000 and 2009. BJU Int 2013;111:E257-62. [Crossref] [PubMed]
- Angioli R, Penalver M, Muzii L, et al. Guidelines of how to manage vesicovaginal fistula. Crit Rev Oncol Hematol 2003;48:295-304. [Crossref] [PubMed]
- Margules AC, Rovner ES. The Use of Tissue Flaps in the Management of Urinary Tract Fistulas. Curr Urol Rep 2019;20:32. [Crossref] [PubMed]
- Stamatakos M, Sargedi C, Stasinou T, et al. Vesicovaginal fistula: diagnosis and management. Indian J Surg 2014;76:131-6. [Crossref] [PubMed]
- Shelbaia AM, Hashish NM. Limited experience in early management of genitourinary tract fistulas. Urology 2007;69:572-4. [Crossref] [PubMed]
- Medina LG, Cacciamani GE, Hernandez A, et al. Robotic Management of Rectourethral Fistulas After Focal Treatment for Prostate Cancer. Urology 2018;118:241. [Crossref] [PubMed]
- Cacciamani GE, Shakir A, Tafuri A, et al. Best practices in near-infrared fluorescence imaging with indocyanine green (NIRF/ICG)-guided robotic urologic surgery: a systematic review-based expert consensus. World J Urol 2020;38:883-96. [Crossref] [PubMed]
- Lee Z, Moore B, Giusto L, et al. Use of indocyanine green during robot-assisted ureteral reconstructions. Eur Urol 2015;67:291. [Crossref] [PubMed]
- Pathak RA, Hemal AK. Intraoperative ICG-fluorescence imaging for robotic-assisted urologic surgery: current status and review of literature. Int Urol Nephrol 2019;51:765-71. [Crossref] [PubMed]
- Vegas L, Sánchez A, Rodríguez O, et al. Sutura y anudado laparoscópico asistido por robot: estudio comparativo de la curva de aprendizaje. Vitae 2013;56:1-7.
- Taylor GW, Jayne DG. Robotic applications in abdominal surgery: their limitations and future developments. Int J Med Robot 2007;3:3-9. [Crossref] [PubMed]
- Vitiello V, Lee SL, Cundy TP, et al. Emerging robotic platforms for minimally invasive surgery. IEEE Rev Biomed Eng 2013;6:111-26. [PubMed]
- Tatar B, Oksay T, Selcen Cebe F, et al. Management of vesicovaginal fistulas after gynecologic surgery. Turk J Obstet Gynecol 2017;14:45-51. [PubMed]
- Boateng AA, Eltahawy EA, Mahdy A. Vaginal repair of ureterovaginal fistula may be suitable for selected cases. Int Urogynecol J 2013;24:921-4. [PubMed]
- Tonolini M. Elucidating vaginal fistulas on CT and MRI. Insights Imaging 2019;10:123. [PubMed]
- Goodwin WE, Scardino PT. Vesicovaginal and ureterovaginal fistulas: a summary of 25 years of experience. J Urol 1980;123:370-4. [PubMed]
- Pietersma CS, Schreuder HW, Kooistra A, et al. Robotic-assisted laparoscopic repair of a vesicovaginal fistula: a time-consuming novelty or an effective tool? BMJ Case Rep 2014;2014:bcr2014204119. [Crossref] [PubMed]
- Randazzo M, Lengauer L, Rochat CH, et al. Best Practices in Robotic-assisted Repair of Vesicovaginal Fistula: A Consensus Report from the European Association of Urology Robotic Urology Section Scientific Working Group for Reconstructive Urology. Eur Urol 2020;78:432-42. [PubMed]
- Chen YB, Wolff BJ, Kenton KS, et al. Approach to Ureterovaginal Fistula: Examining 13 Years of Experience. Female Pelvic Med Reconstr Surg 2019;25:e7-e11. [PubMed]
- Kwon EO, Armenakas NA, Scharf SC, et al. The poppy seed test for colovesical fistula: big bang, little bucks! J Urol 2008;179:1425-7. [PubMed]
- Garcea G, Majid I, Sutton CD, et al. Diagnosis and management of colovesical fistulae; six-year experience of 90 consecutive cases. Colorectal Dis 2006;8:347-52. [PubMed]
- La Riva A, Perez LC, Sayegh AS, et al. Fistulae: vesicovaginal, ureterovaginal, and rectourethral. Principles and Practice of Robotic Surgery. Costello 2020:31-58 .
- Medina LG, Rangel E, Fuchs I, et al. Rectourethral fistula: operative technique and outcomes. Curr Bladder Dysfunct Rep 2019;14:151-6.
- Bodner-Adler B, Hanzal E, Pablik E, et al. Management of vesicovaginal fistulas (VVFs) in women following benign gynaecologic surgery: A systematic review and meta-analysis. PLoS One 2017;12:e0171554. [PubMed]
- Miklos JR, Moore RD, Chinthakanan O. Laparoscopic and Robotic-assisted Vesicovaginal Fistula Repair: A Systematic Review of the Literature. J Minim Invasive Gynecol 2015;22:727-36. [Crossref] [PubMed]
- Núñez Bragayrac LA, Azhar RA, Sotelo R. Minimally invasive management of urological fistulas. Curr Opin Urol 2015;25:136-42. [Crossref] [PubMed]
- Sotelo R, Mariano MB, García-Segui A, et al. Laparoscopic repair of vesicovaginal fistula. J Urol 2005;173:1615-8. [PubMed]
- Eilber KS, Kavaler E, Rodríguez LV, et al. Ten-year experience with transvaginal vesicovaginal fistula repair using tissue interposition. J Urol 2003;169:1033-6. [Crossref] [PubMed]
- Martínez JÁ, Hernández-Castellanos V, Fernández-Noyola G, et al. Fístulas vesico-vaginales; diagnóstico y manejo; experiencia de 20 años. Rev Mex Urol 2011;71:200-6.
- Matei DV, Zanagnolo V, Vartolomei MD, et al. Robot-Assisted Vesico-Vaginal Fistula Repair: Our Technique and Review of the Literature. Urol Int 2017;99:137-42. [Crossref] [PubMed]
- Sayegh AS, La Riva A, Perez LC, et al. Robotic-assisted vesicovaginal fistula repair using a vaginal cuff flap. Int Urogynecol J 2022;33:2581-5. [Crossref] [PubMed]
- La Riva A, Perez LC, Sayegh AS, et al. Robotic Repair of Vesicovaginal Fistula: Step-by-Step Technique for Complex Scenarios. Videourology 2021;35(8). Available online:
10.1089/vid.2021.0036 10.1089/vid.2021.0036 - Rogers RG, Jeppson PC. Current Diagnosis and Management of Pelvic Fistulae in Women. Obstet Gynecol 2016;128:635-50. [Crossref] [PubMed]
- Serna-Gallegos T, Jeppson PC. Female Pelvic Fistulae. Obstet Gynecol Clin North Am 2021;48:557-70. [Crossref] [PubMed]
- Garza Cortez R, Clavijo RS. Tratamiento Laparoscópico de Fístulas Genitourinarias. Arch Esp Urol 2012;65:659-72.
- Dutto L, O'Reilly B. Robotic repair of vesico-vaginal fistula with perisigmoid fat flap interposition: state of the art for a challenging case? Int Urogynecol J 2013;24:2029-30. [Crossref] [PubMed]
- Hemal AK, Kolla SB, Wadhwa P. Robotic reconstruction for recurrent supratrigonal vesicovaginal fistulas. J Urol 2008;180:981-5. [Crossref] [PubMed]
- Price DT, Price TC. Robotic repair of a vesicovaginal fistula in an irradiated field using a dehydrated amniotic allograft as an interposition patch. J Robot Surg 2016;10:77-80. [PubMed]
- Sharma AP, Mavuduru RM, Bora GS, et al. Robot-assisted Vesico-vaginal Fistula Repair: A Compilation. Urology 2018;119:1-4. [PubMed]
- Kelly E, Wu MY, MacMillan JB. Robotic-assisted vesicovaginal fistula repair using an extravesical approach without interposition grafting. J Robot Surg 2018;12:173-6. [PubMed]
- Kurz M, Horstmann M, John H. Robot-assisted laparoscopic repair of high vesicovaginal fistulae with peritoneal flap inlay. Eur Urol 2012;61:229-30. [PubMed]
- Dalela D, Ranjan P, Sankhwar PL, et al. Supratrigonal VVF repair by modified O'Connor's technique: an experience of 26 cases. Eur Urol 2006;49:551-6. [PubMed]
- O'Conor VJ, Sokol JK. Vesicovaginal fistula from the standpoint of the urologist. J Urol 1951;66:579-85. [PubMed]
- Nesrallah LJ, Srougi M, Gittes RF. The O'Conor technique: the gold standard for supratrigonal vesicovaginal fistula repair. J Urol 1999;161:566-8. [PubMed]
- Occhino JA, Hokenstad ED, Linder BJ. Robot-assisted vesicovaginal fistula repair via a transvesical approach. Int Urogynecol J 2019;30:327-9. [PubMed]
- Gupta NP, Mishra S, Hemal AK, et al. Comparative analysis of outcome between open and robotic surgical repair of recurrent supra-trigonal vesico-vaginal fistula. J Endourol 2010;24:1779-82. [PubMed]
- Kumar A, Goyal NK, Das SK, et al. Our experience with genitourinary fistulae. Urol Int 2009;82:404-10. [Crossref] [PubMed]
- Bora GS, Singh S, Mavuduru RS, et al. Robot-assisted vesicovaginal fistula repair: a safe and feasible technique. Int Urogynecol J 2017;28:957-62. [Crossref] [PubMed]
- Bettez M, Breault G, Carr L, et al. Early versus delayed repair of vesicouterine fistula. Can Urol Assoc J 2011;5:E52-5. [Crossref] [PubMed]
- Baker MV, Kisby CK, Occhino JA. Vesicouterine Fistula: A Robotic Approach. Int Urogynecol J 2022;33:1685-7. [Crossref] [PubMed]
- Rajamaheswari N, Chhikara AB. Vesicouterine fistulae: our experience of 17 cases and literature review. Int Urogynecol J 2013;24:275-9. [Crossref] [PubMed]
- Purkait B, Mehrotra S, Sinha RJ, et al. Long-term outcome of laparoscopic vesicouterine fistula repair: Experience from a tertiary referral centre. Turk J Urol 2017;43:512-6. [Crossref] [PubMed]
- Symeonidis EN, Sdralis E, Symeonidis A, et al. Vesicouterine Fistula (VUF) as a Rare Urogenital Complication Managed with Delayed Surgical Repair: A Case Report and Review of the Literature. Case Rep Obstet Gynecol 2018;2018:2394896. [Crossref] [PubMed]
- Józwik M, Józwik M. Clinical classification of vesicouterine fistula. Int J Gynaecol Obstet 2000;70:353-7. [Crossref] [PubMed]
- Hemal AK, Sharma N, Mukherjee S. Robotic repair of complex vesicouterine fistula with and without hysterectomy. Urol Int 2009;82:411-5. [Crossref] [PubMed]
- Hemal AK, Kumar R, Nabi G. Post-cesarean cervicovesical fistula: technique of laparoscopic repair. J Urol 2001;165:1167-8. [Crossref] [PubMed]
- Li X, Wang P, Liu Y, et al. Minimally invasive surgical treatment on delayed uretero-vaginal fistula. BMC Urol 2018;18:96. [Crossref] [PubMed]
- Ramírez-Martín D, Jara-Rascón J, Renedo-Villar T, et al. Rectourethral Fistula Management. Curr Urol Rep 2016;17:22. [Crossref] [PubMed]
- Shaw J, Tunitsky-Bitton E, Barber MD, et al. Ureterovaginal fistula: a case series. Int Urogynecol J 2014;25:615-21. [Crossref] [PubMed]
- Bragayrac LA, Azhar RA, Fernandez G, et al. Robotic repair of vesicovaginal fistulae with the transperitoneal-transvaginal approach: a case series. Int Braz J Urol 2014;40:810-5. [Crossref] [PubMed]
- Kidd LC, Lee M, Lee Z, et al. A Multi-Institutional Experience with Robotic Vesicovaginal and Ureterovaginal Fistula Repair After Iatrogenic Injury. J Endourol 2021;35:1659-64. [Crossref] [PubMed]
- Gellhaus PT, Bhandari A, Monn MF, et al. Robotic management of genitourinary injuries from obstetric and gynaecological operations: a multi-institutional report of outcomes. BJU Int 2015;115:430-6. [Crossref] [PubMed]
- Zinman LM, Libertino JA, Roth RA. Management of operative ureteral injury. Urology 1978;12:290-303. [PubMed]
- Ehrlich RM, Melman A, Skinner DG. The use of vesico-psoas hitch in urologic surgery. J Urol 1978;119:322-5. [PubMed]
- Barone MA, Widmer M, Arrowsmith S, et al. Breakdown of simple female genital fistula repair after 7 day versus 14 day postoperative bladder catheterisation: a randomised, controlled, open-label, non-inferiority trial. Lancet 2015;386:56-62. [PubMed]
- Torloni MR, Riera R, Rogozińska E, et al. Systematic review of shorter versus longer duration of bladder catheterization after surgical repair of urinary obstetric fistula. Int J Gynaecol Obstet 2018;142:15-22. [PubMed]
- Helfand AM, Beach R, Hadj-Moussa M, et al. Treatment of ureteral anastomotic strictures with reimplantation and survival after cystectomy and urinary diversion. Urol Oncol 2017;35:33.e1-33.e9. [PubMed]
- Mellano EM, Tarnay CM. Management of genitourinary fistula. Curr Opin Obstet Gynecol 2014;26:415-23. [PubMed]
Cite this article as: Rangel EM, Morocco E, Sotelo R. Robotic surgery for urinary fistulae in females: a narrative review. Gynecol Pelvic Med 2025;8:1.





